Insights into the structure of a protein transport assistant
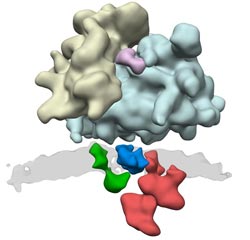
Many protein complexes are involved in protein synthesis.Through the ER translocon (green, blue and red) the newly synthesized protein is transported across the membrane (gray).<br><br>Graphic: Friedrich Förster / Copyright: MPI of Biochemistry<br>
After protein production, many proteins are equipped with attachments such as sugar residues in order to perform their tasks properly. This process is directly coupled to the transport across a membrane.
Employing various methods of structural biology, scientists at the Max Planck Institute (MPI) of Biochemistry in Martinsried near Munich, Germany, have now gained insights into the architecture of the protein complex (ER translocon) responsible for this process. The results of the joint project have now been published in Nature Communications.
Producing a protein is a highly intricate process for the cell and involves many individual steps. Depending on the purpose for which a protein is used, there are different sites for protein production: the cytoplasm or the endoplasmic reticulum (ER). The ER is separated by a membrane from its surroundings in the cytoplasm. Even before protein synthesis is completed, the proteins produced at the ER enter via its membrane into the interior of the ER and are modified through the attachment of sugar residues concomitantly. Without these attachments, the proteins would not be able to fold properly and thus would not fulfill their functions in the cell.
Scientists of the research group “Modeling of Protein Complexes” have now described the architecture of the protein complex responsible for the transport and modification of the newly produced protein: the ER translocon. “It is located in the membrane of the ER, and this fact, together with its size and complex composition, has greatly hampered previous structural studies,” says Friedrich Förster, group leader at the MPI of Biochemistry, describing the initial situation. The structures of many subunits and their arrangement in the native ER translocon have thus far remained elusive.
It was not until cryoelectron tomography came into use that researchers could gain first insight into the architecture of the translocon. The sample is “shock frozen” to preserve its natural structure. Using an electron microscope, the scientists capture two-dimensional images of the object from different perspectives, from which they then reconstruct a three-dimensional image. Further investigations have made it possible to identify individual modules in the structure. Among them is the module that attaches the sugar residues to the newly produced protein.
“Based on this method, we will now try to determine the structure and location of other components of the ER translocon,” says Förster. If the researchers know the individual structures of the ER translocon and their arrangement in the complex, they can indirectly draw conclusions about the precise functions and interactions of all components.
Original publication
Pfeffer, S., Dudek, J., Gogala, M., Schorr, S., Linxweiler, J., Lang, S., Becker, T., Beckmann, R., Zimmermann, R., Förster, F.: Structure of the mammalian oligosaccharyl-transferase complex in the native ER protein translocon. Nature Commun, January 10, 2014
Doi: 10.1038/ncomms4072 (2013).
Contact
Dr. Friedrich Förster
Modeling of Protein Complexes
Max Planck Institute of Biochemistry
Am Klopferspitz 18
82152 Martinsried
Germany
Email: foerster@biochem.mpg.de
http://www.biochem.mpg.de/foerster
Anja Konschak
Public Relations
Max Planck Institute of Biochemistry
Am Klopferspitz 18
82152 Martinsried
Germany
Tel. +49 89 8578-2824
E-Mail: konschak@biochem.mpg.de
http://www.biochem.mpg.de
Weitere Informationen:
http://www.biochem.mpg.de/foerster
– website of the research group “Modeling of Protein Complexes” (Friedrich Förster)
http://www.biochem.mpg.de/3686257/071_foerster_translocon_nature
– complete press release
Media Contact
More Information:
http://www.biochem.mpg.deAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Making diamonds at ambient pressure
Scientists develop novel liquid metal alloy system to synthesize diamond under moderate conditions. Did you know that 99% of synthetic diamonds are currently produced using high-pressure and high-temperature (HPHT) methods?[2]…

Eruption of mega-magnetic star lights up nearby galaxy
Thanks to ESA satellites, an international team including UNIGE researchers has detected a giant eruption coming from a magnetar, an extremely magnetic neutron star. While ESA’s satellite INTEGRAL was observing…

Solving the riddle of the sphingolipids in coronary artery disease
Weill Cornell Medicine investigators have uncovered a way to unleash in blood vessels the protective effects of a type of fat-related molecule known as a sphingolipid, suggesting a promising new…





















