Airborne Particles Smuggle Pollutants to Far Reaches of Globe
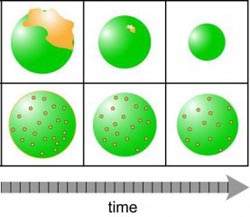
Zelenyuk/PNNL<br> <br>When airborne particles (green) form before pollutants known as PAHs (yellow) glob on, both the pollutants and particles dissipate quickly, as shown in the top row. But when the particles form in the presence of pollutants, which is what likely happens in nature, the long-lasting particles can take the pollutants for a long-distance ride (bottom)<br>
Pollution from fossil fuel burning and forest fires reaches all the way to the Arctic, even though it should decay long before it travels that far. Now, lab research can explain how pollution makes its lofty journey: rather than ride on the surface of airborne particles, pollutants snuggle inside, protected from the elements on the way. The results will help scientists improve atmospheric air-quality and pollution transport models.
The results also show that the particles that envelop pollutants also benefit from this arrangement. The new study in Environmental Science & Technology shows that the airborne particles, made from natural molecules mostly given off by live or burning plants, last longer with a touch of pollutant packed inside. The pollutants are known as polycyclic aromatic hydrocarbons, or PAHs, and are regulated by environmental agencies due to their toxicity.
“What we've learned through fundamental studies on model systems in the lab has very important implications for long-range transport of pollutants in the real world,” said physical chemist Alla Zelenyuk of the Department of Energy's Pacific Northwest National Laboratory. “In this study, we propose a new explanation for how PAHs get transported so far, by demonstrating that airborne particles become a protective vessel for PAH transport.”
Floating in the air and invisible to the eye, airborne particles known as secondary organic aerosols live and die. Born from carbon-based molecules given off by trees, vegetation, and fossil fuel burning, these airborne SOA particles travel the currents and contribute to cloud formation. Along for the ride are pollutants, the PAHs, that have long been thought to coat the particles on their surface.
For decades, atmospheric scientists have been trying to explain how atmospheric particles manage to transport harmful pollutants to pristine environments thousands of miles away from their starting point. The particles collected in areas such as the Arctic also pack higher concentrations of pollutants than scientists' computer models predict.
The predictions are based on the assumption that the particles are like liquid spheres, whose fluidity allows PAHs to escape. But they don't escape, and one recent advance has helped to pin down why PAHs are remaining stuck in their particle lairs. Zelenyuk and her colleagues at EMSL, DOE's Environmental Molecular Sciences Laboratory at PNNL, developed an ultra-sensitive instrument that can determine the size, composition and shape of individual particles.
Called SPLAT II, the instrument can analyze millions of tiny particles one by one. The ability of this novel instrument to characterize individual particles provides unique insight into their property and evolution.
Using SPLAT II to evaluate laboratory-generated SOA particles from alpha-pinene, the molecule that gives pine trees their piney smell, Zelenyuk has already discovered that SOA particles aren't liquid at all. Her team's recent work revealed they are more like tar — thick, viscous blobs that are too solid to be liquid and too liquid to be solid.
Armed with this data, Zelenyuk and researchers from Imre Consulting in Richland and the University of Washington in Seattle set out to determine the relation between the SOA particle and the PAHs. Again they used alpha-pinene for the SOA. For the PAH, they used pyrene, a toxic pollutant produced by burning fossil fuels or vegetation such as forests.
They created two kinds of particles. The first kind exemplified the classical SOA: first they produced the particles with alpha-pinene and then coated them with pyrene. The second kind resembled what likely happens in nature: they mixed alpha-pinene and pyrene and let the particles form with both molecules present. Then they sent the particles through SPLAT and watched what happened to them over time.
With the pyrene-coated particles, the team found the PAH pyrene evaporating off the surface of the particle quickly, all of it gone after four hours. By the next day, the particle itself had shrunk by about 70 percent, showing that the alpha-pinene SOA also evaporates, although more slowly than pyrene.
When they created the particles in the presence of both SOA and PAH, the PAH evaporated much more slowly. Fifty percent of the original PAH still remained in the particle after 24 hours. In addition, the SOA particle itself stayed bulky, losing less than 20 percent of its volume.
These results showed the team that PAHs become trapped within the highly viscous SOA particles, where they remain protected from the environment. The symbiotic relationship between the atmospheric particles and pollutants surprised Zelenyuk: SOAs help PAHs travel the world, and the PAHs help SOAs survive longer.
Zelenyuk and her colleagues performed comparable experiments with other PAHs and SOAs and found similar results.
In the real world, Zelenyuk said, the evaporation will be even slower. These results will help modelers better simulate atmospheric SOA particles and transport of pollutants over long distances.
This work was supported by the Department of Energy Office of Science and PNNL's Chemical Imaging Initiative.
Reference: Alla Zelenyuk, Dan Imre, Josef Beránek, Evan Abramson, Jacqueline Wilson and Manish Shrivastava, Synergy between Secondary Organic Aerosols and Long-Range Transport of Polycyclic Aromatic Hydrocarbons, Environmental Science & Technology, Nov. 7, 2012, doi: 10.1021/es302743z. (http://pubs.acs.org/doi/abs/10.1021/es302743z)
EMSL, the Environmental Molecular Sciences Laboratory, is a national scientific user facility sponsored by the Department of Energy's Office of Science. Located at Pacific Northwest National Laboratory in Richland, Wash., EMSL offers an open, collaborative environment for scientific discovery to researchers around the world. Its integrated computational and experimental resources enable researchers to realize important scientific insights and create new technologies. Follow EMSL on Facebook, LinkedIn and Twitter.
Interdisciplinary teams at Pacific Northwest National Laboratory address many of America's most pressing issues in energy, the environment and national security through advances in basic and applied science. PNNL employs 4,500 staff, has an annual budget of nearly $1 billion, and has been managed for the U.S. Department of Energy by Ohio-based Battelle since the laboratory's inception in 1965. For more, visit the PNNL's News Center, or follow PNNL on Facebook, LinkedIn and Twitter.
DOE’s Office of Science is the single largest supporter of basic research in the physical sciences in the United States, and is working to address some of the most pressing challenges of our time. For more information, please visit science.energy.gov.
Media Contact
More Information:
http://www.pnnl.govAll latest news from the category: Ecology, The Environment and Conservation
This complex theme deals primarily with interactions between organisms and the environmental factors that impact them, but to a greater extent between individual inanimate environmental factors.
innovations-report offers informative reports and articles on topics such as climate protection, landscape conservation, ecological systems, wildlife and nature parks and ecosystem efficiency and balance.
Newest articles

Peptides on Interstellar Ice
A research team led by Dr Serge Krasnokutski from the Astrophysics Laboratory at the Max Planck Institute for Astronomy at the University of Jena had already demonstrated that simple peptides…

A new look at the consequences of light pollution
GAME 2024 begins its experiments in eight countries. Can artificial light at night harm marine algae and impair their important functions for coastal ecosystems? This year’s project of the training…

Silicon Carbide Innovation Alliance to drive industrial-scale semiconductor work
Known for its ability to withstand extreme environments and high voltages, silicon carbide (SiC) is a semiconducting material made up of silicon and carbon atoms arranged into crystals that is…





















